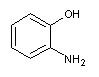
o-aminophenol
structural formula
| business number | 029c |
|---|---|
| molecular formula | c6h7no |
| molecular weight | 109 |
| label |
o-hydroxyaniline, 2-aminophenol, 1-amino-2-hydroxybenzene, 2-amino-1-hydroxybenzene, o-aminophenol, 2-aminophenol, aromatic nitrogen-containing compounds and their derivatives |
numbering system
cas number:95-55-6
mdl number:mfcd00007690
einecs number:202-431-1
rtecs number:sj4950000
brn number:606075
pubchem number:24891176
physical property data
1. properties: white or light gray crystalline powder. [1]
2. melting point (℃): 170~174[2]
3. octanol/water distribution coefficient: 0.52~0.62[3]
4. solubility: soluble in cold water, ethanol, benzene, and ether. [4]
toxicological data
1. acute toxicity[5]
ld50: 951mg/kg (rat oral); 800mg/kg (mouse oral oral); >1g/kg (guinea pig transdermal)
2. irritation [6] rabbit eye: 100mg, mild irritation .
3. mutagenicity [7] microbial mutagenicity: salmonella typhimurium 333μg/dish.
4. teratogenicity[8] the lowest toxic dose (tdlo) of 150mg/kg was given intraperitoneally to hamsters on the 8th day after pregnancy, causing damage to the central nervous system. , eyes, ears, and musculoskeletal system developmental malformations. dna inhibition: hamster lung 19 μmol/l.
ecological data
1. ecotoxicity no data yet
2. biodegradability[9] when the cod value of 2-aminophenol is 200 mg/l, domesticated activated sludge is used, and the sludge concentration reaches 100 mg/l. after 5 days (aerobic), the cod removal rate is 95%.
3. non-biodegradability[10] in the air, when the hydroxyl radical concentration is 5.00×105 pcs/cm3, the degradation half-life is 5h (theoretical).
molecular structure data
1. molar refractive index: 32.37
2. molar volume (cm3/mol): 90.1
3. isotonic specific volume (90.2k ): 248.1
4. surface tension (dyne/cm): 57.4
5. polarizability (10-24cm3): 12.83
compute chemical data
1. reference value for hydrophobic parameter calculation (xlogp): none
2. number of hydrogen bond donors: 2
3. number of hydrogen bond acceptors: 2
4. number of rotatable chemical bonds: 0
5. number of tautomers: 7
6. topological molecule polar surface area 46.2
7. number of heavy atoms: 8
8. surface charge: 0
9. complexity: 74.9
10. number of isotope atoms: 0
11. determine the number of atomic stereocenters: 0
12. uncertain number of atomic stereocenters: 0
13. determined number of chemical bond stereocenters: 0
14. uncertain number of chemical bond stereocenters: 0
15. number of covalent bond units: 1
properties and stability
1. stability[11] stable
2. incompatible substances[12] strong oxidants, acid chlorides, acid anhydrides, acids, chloroform
3. conditions to avoid contact [13] heat
>
4. hazards of aggregation[14] no aggregation
storage method
storage precautions[15] store in a cool, ventilated warehouse. keep away from fire and heat sources. the packaging is sealed. they should be stored separately from oxidants, acids, and food chemicals, and avoid mixed storage. equipped with the appropriate variety and quantity of fire equipment. suitable materials should be available in the storage area to contain spills.
synthesis method
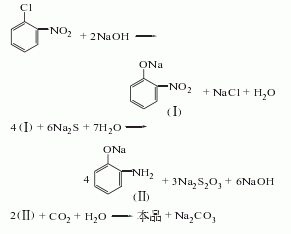
heat o-nitrochlorobenzene in a sodium hydroxide solution and hydrolyze it under a certain pressure to obtain sodium o-nitrophenolate, which is then reduced with alkali sulfide. then use carbonic acid to separate, cool, filter, soak in sodium bisulfite solution, centrifuge, and dry to obtain the finished product.
purpose
1. dye intermediates, used to manufacture sulfur dyes, azo dyes, fur dyes and fluorescent whitening agent eb, etc. raw materials used in the pesticide industry for the pesticide phosphos.
2. mainly used to make acid medium blue r, sulfur yellow brown, etc., and can also be used as fur dye. used in the cosmetics industry to make hair dyes (as compound dyes).
3. determination of silver and tin and verification of gold. intermediate of diazo dyes and sulfur dyes.
4. used in the manufacture of dyes, drugs, and plastic curing agents. [16]
extended-reading:https://www.bdmaee.net/246-trisdimethylaminomethylphenol/extended-reading:https://www.morpholine.org/dabco-33-s-microporous-catalyst/extended-reading:https://www.bdmaee.net/high-efficiency-catalyst-pt303/extended-reading:https://www.bdmaee.net/fomrez-ul-29-catalyst-octylmercaptan-stannous–2/extended-reading:https://www.bdmaee.net/cas-818-08-6-3/extended-reading:https://www.newtopchem.com/archives/45041extended-reading:https://www.newtopchem.com/archives/44147extended-reading:https://www.bdmaee.net/wp-content/uploads/2022/08/newtop7.jpgextended-reading:https://www.newtopchem.com/archives/category/products/page/22extended-reading:https://www.bdmaee.net/dabco-b-16-amine-catalyst-b16-dabco-b16/

